BrosMed’s POT™ NC PTCA Balloon Dilatation Catheter Receives FDA 510(k) Clearance
April 12, 2023—BrosMed Medical has received FDA 510(k) clearance for the POT™ NC PTCA Balloon Dilatation Catheter, which is the world’s first dedicated balloon for proximal optimization technique (POT) and distal optimization technique (DOT) in bifurcation stenting. With this certification, a specialized tool in the treatment of precise PCI for bifurcation lesions is available to US physicians.

Coronary bifurcation accounts for 15-20% of all percutaneous coronary interventions (PCI), which is still expected to be one of the most challenging lesions in interventional cardiology in terms of a low procedural success rate as well as long-term cardiac events.
The proximal optimization technique (POT) has been proposed as an effective strategy to achieve optimal stent expansion and apposition in the proximal segment in coronary bifurcation. It is a straightforward technique whereby a short, appropriately-sized balloon like POT™ is inflated in the main vessel just proximal to the carina. The advantages of this technique include reducing the risk of side branch compromise related to shifting of the carina, improving stent apposition in the proximal main vessel, as well as facilitating side branch access after main vessel stent implantation.
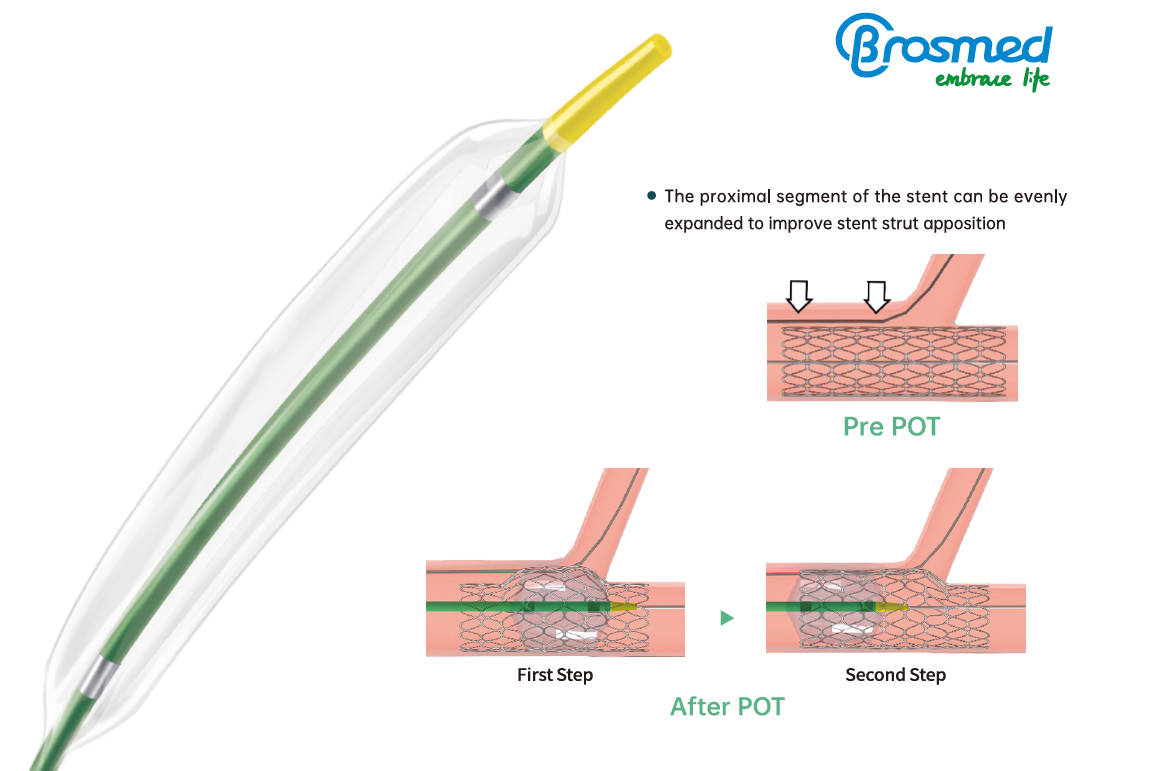
As the special-designed balloon for POT and DOT technique, POT™ reigns as the best choice of treating bifurcations undoubtedly. The POT™ PTCA catheter is a non-compliant balloon designed with extra short balloon shoulders and precise apposition performance in order to reduce longitudinal balloon growth and minimize the potential for vessel trauma outside the treatment area in bifurcation stenting.
Prof. Shao-Liang Chen, the inventor of DK-crush, “A real POT™ balloon innovated by BrosMed has provided the final solution for either POT or DOT. POT™ PTCA balloon catheter offers tremendous promise and adds a state-of-the-art coronary balloon to BrosMed leading products portfolio.”
BrosMed will strive to continue developing innovative devices to address the most complicated clinical challenges and make contributions to physicians and patients worldwide, as well as the development of the medical industry.
For more information about POT™ NC PTCA, please visit BrosMed website or contact us via e-mail: Sales@brosmed.com.

